 
Image showing periodicity of the chemical elements for atomic radius (empirical) in a 3D spiral periodic table column style. Image showing periodicity of the chemical elements for atomic radius (empirical) in a spiral periodic table heatscape style. The reasons for grouping Be, Mg and Ca into IIA are as follows. 
Atomic Radius Trend 2: Atomic radii increases while moving to a group the second periodic tendency of the atomic radius is that atomic radii increases while moving down to a group in the periodic table. Image showing periodicity of the chemical elements for atomic radius (empirical) in a periodic table heatscape style. And this is true Carbon has an average atomic radius of about 70 pm while fluoride is 50 in the morning. Image showing periodicity of the chemical elements for atomic radius (empirical) in a periodic table cityscape style. Write the electron configurations for a) Ca b) Co c)S 5. Referring to the periodic table, arrange the atoms Ne, Na, P, Ar, K in order of increasing first ionization energy. Arrange Mg, Car, and Ca in order of decreasing radius 3. 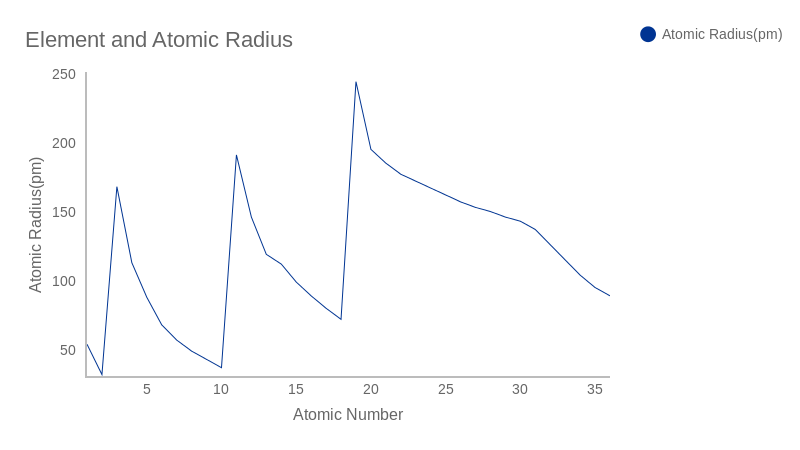
Values are given to an accuracy of about 5 pm. Arrange Be, C, K, and Ca in order of increasing atomic radius 2. A single set of radii is very useful for most purposes, however, for very accurate work adjustments would have to be made to the values quoted to reflect the specific environment of the element (such as coordination number). This is due to trends in the periodic table, and the effective nuclear charge that holds the valence electrons close to. Solve any question of Classification Of Elements And Periodicity In Properties with:. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. Up to date, curated data provided by Mathematica 's ElementData function from Wolfram Research, Inc. Correct option is C) On moving down the group atomic radius increases, therefore the decreasing order comes out to be Ra > Ba > Sr > Ca > Mg > Be. ionic radii of group 1 cations increases down the group. It must be noted, atoms lack a well-defined outer boundary. ionic radius of an anion is greater than atomic radius of the atom. The idea is that for a bond A-B, the atomic radius of A added to the atomic radius of B will give a reasonable estimate for the A-B separation in whatever environment. The atomic radius of Calcium atom is 176pm (covalent radius). Slater are an empirical set of atomic radii derived by the careful comparison of bond lengths in over 1200 bond types in ionic, metallic, and covalent crystals and molecules (reference 1). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
